In the field of organic chemistry, functional groups are the substituent atoms or groups of atoms that are bound to specific molecules. Such moieties are part of the molecule that can be present in many other molecules due to chemical reactions.
Two molecules of different sizes with the same functional groups will participate in approximately the same chemical reactions. The presence of a functional group in a molecule involves a systematic prediction and the chemical reactions of the molecule. General examples of functional examples are amine, alcohols, carboxylic acids, ethers, aldehydes and ketones.
Organic Chemistry Functional Groups
Amines
Amine is a kind of substance derived from ammonia (NH3), or amines are ammonia derivatives. They are generally known as functional groups or organic compounds of nitrogen which contain a lone pair of nitrogen atoms. Within amines, hydrogen atoms are usually substituted by an aryl or alkyl group. Various types of amines are primary amines, secondary amines, tertiary amines and quaternary ammonium salt.
Alcohols
Alcohols are those chemical compounds that are distinguished by the presence of one, two or more OH groups (hydroxyl groups) in the hydrocarbon chain or alkyl group connected to the carbon atom. Some of the most commonly found organic compounds are considered to be alcohols. They are used in the form of sweeteners, fragrance preparation, and also in the cycle of synthesizing certain substances, in which certain compounds are abundantly generated in organic compounds coming from different sectors. Different types of alcoholic compounds are 10, 20 and 30 alcohols.
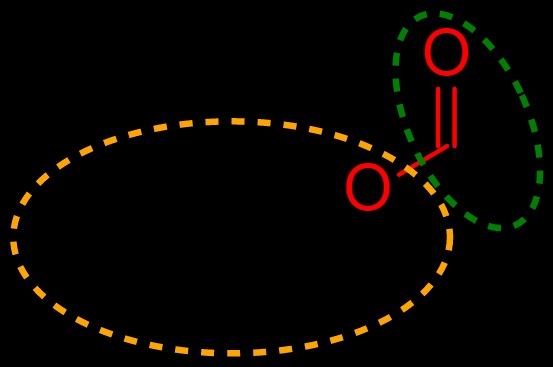
Carboxylic acid
A Carboxylic Acid is an organic compound that comprises a functional group of carboxyls; they are commonly found in nature and are also produced synthetically by humankind. Carboxylic acids yield a carboxylate anion with the general formula R-COO– after deprotonation. The carboxylate ions can form several useful salts, including soaps.
The most important functional group representing C = O is the carboxylic acids. These types of chemical compounds could be generated by various paths, some carboxylic acids, including certain lactic acid, citric acid or fumaric acid, are formed by fermentation and most of these carboxylic acids are used in the food sector.
Aldehydes and Ketones
Aldehydes and ketones contain a functional group of carbonyls, C = O. These are chemical molecules with structures -CHO and RC(=O)R ‘where R and R’ represent substituents that contain carbon, respectively.
Examples
- CH3 – CO – H – Ethanal
- CH3 – CO – CH, – Propanone
Ethers
Ethers are a class of organic compounds that come from the Latin term ‘aether,’ meaning ‘to ignite, it is flammable at room temperature, under high pressure. In the ethers oxygen atom, bound to two alkyl or aryl groups equivalent or distinct. The Ether’s general formula is given as R-O-R, R-O-R’, R-O-Ar or Ar-O-Ar where R represents an alkyl group and Ar is an aryl group.
Examples:
- (CH3)2CH – O – CH(CH3)2 – Diisopropyl Ether
- C6H5 – O – C6H5 – Diphenyl ether
Conclusion
The functional group plays a significant role in the field of chemistry. The functional groups that participate in a chemical reaction can be further modified with the help of other functional groups, and these groups can also be interconverted. It can, therefore, be recognized that functional groups are the moieties that possess their distinct characteristics and properties, independent of the molecule to which they are attached.